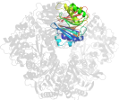
Lineage for d1ecjd2 (1ecj D:1-249)
- Root: SCOPe 2.08

Class d: Alpha and beta proteins (a+b) [53931] (396 folds) 
Fold d.153: Ntn hydrolase-like [56234] (2 superfamilies)
4 layers: alpha/beta/beta/alpha; has an unusual sheet-to-sheet packing
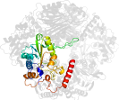
Superfamily d.153.1: N-terminal nucleophile aminohydrolases (Ntn hydrolases) [56235] (8 families) 
N-terminal residue provides two catalytic groups, nucleophile and proton donor
Family d.153.1.1: Class II glutamine amidotransferases [56236] (6 proteins)
has slightly different topology than other families do
Protein Glutamine PRPP amidotransferase, N-terminal domain [56239] (3 species) 
Species Escherichia coli [TaxId:562] [56241] (5 PDB entries) 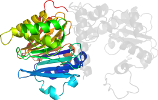
Domain d1ecjd2: 1ecj D:1-249 [41829]
Other proteins in same PDB: d1ecja1, d1ecjb1, d1ecjc1, d1ecjd1
complexed with amp
Details for d1ecjd2
PDB Entry: 1ecj (more details), 2.5 Å
PDB Description: escherichia coli glutamine phosphoribosylpyrophosphate (prpp) amidotransferase complexed with 2 amp per tetramer
PDB Compounds: (D:) glutamine phosphoribosylpyrophosphate amidotransferaseSCOPe Domain Sequences for d1ecjd2:
Sequence; same for both SEQRES and ATOM records: (download)
>d1ecjd2 d.153.1.1 (D:1-249) Glutamine PRPP amidotransferase, N-terminal domain {Escherichia coli [TaxId: 562]}
cgivgiagvmpvnqsiydaltvlqhrgqdaagiitidanncfrlrkanglvsdvfearhm
qrlqgnmgighvryptagsssaseaqpfyvnspygitlahngnltnahelrkklfeekrr
hinttsdseillnifaseldnfrhypleadnifaaiaatnrlirgayacvamiighgmva
frdpngirplvlgkrdidenrteymvasesvaldtlgfdflrdvapgeaiyiteegqlft
rqcadnpvs
SCOPe Domain Coordinates for d1ecjd2:
Click to download the PDB-style file with coordinates for d1ecjd2.
(The format of our PDB-style files is described here.)
(The format of our PDB-style files is described here.)
Timeline for d1ecjd2:
- d1ecjd2 first appeared (with stable ids) in SCOP 1.55
- d1ecjd2 appears in SCOPe 2.07
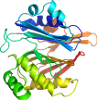
 View in 3D
View in 3D