Lineage for d1l1ye_ (1l1y E:)
- Root: SCOPe 2.08

Class a: All alpha proteins [46456] (290 folds) 
Fold a.102: alpha/alpha toroid [48207] (6 superfamilies)
multihelical; up to seven alpha-hairpins are arranged in closed circular array; there may be sequence similarities between different superfamilies
Superfamily a.102.1: Six-hairpin glycosidases [48208] (10 families) 

Family a.102.1.2: Cellulases catalytic domain [48213] (12 proteins) 
Protein Processive endocellulase CelF (Cel48F) [48220] (2 species) 
Species Clostridium thermocellum [TaxId:1515] [74765] (2 PDB entries) 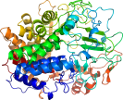
Domain d1l1ye_: 1l1y E: [73490]
has additional subdomain(s) that are not in the common domain
Details for d1l1ye_
PDB Entry: 1l1y (more details), 2.4 Å
PDB Description: the crystal structure and catalytic mechanism of cellobiohydrolase cels, the major enzymatic component of the clostridium thermocellum cellulosome
PDB Compounds: (E:) cellobiohydrolaseSCOPe Domain Sequences for d1l1ye_:
Sequence; same for both SEQRES and ATOM records: (download)
>d1l1ye_ a.102.1.2 (E:) Processive endocellulase CelF (Cel48F) {Clostridium thermocellum [TaxId: 1515]}
gptkaptkdgtsykdlflelygkikdpkngyfspdegipyhsietliveapdyghvttse
afsyyvwleamygnltgnwsgvetawkvmedwiipdsteqpgmssynpnspatyadeyed
psyypselkfdtvrvgsdpvhndlvsaygpnmylmhwlmdvdnwygfgtgtratfintfq
rgeqestwetiphpsieefkyggpngfldlftkdrsyakqwrytnapdaegraiqavywa
nkwakeqgkgsavasvvskaakmgdflrndmfdkyfmkigaqdktpatgydsahylmawy
tawgggigaswawkigcshahfgyqnpfqgwvsatqsdfapkssngkrdwttsykrqlef
yqwlqsaeggiaggatnswngryekypagtstfygmayvphpvyadpgsnqwfgfqawsm
qrvmeyyletgdssvknlikkwvdwvmseiklyddgtfaipsdlewsgqpdtwtgtytgn
pnlhvrvtsygtdlgvagslanalatyaaaterwegkldtkardmaaelvnrawynfycs
egkgvvteearadykrffeqevyvpagwsgtmpngdkiqpgikfidirtkyrqdpyydiv
yqaylrgeapvlnyhrfwhevdlavamgvlatyfpdmtykvp
SCOPe Domain Coordinates for d1l1ye_:
Click to download the PDB-style file with coordinates for d1l1ye_.
(The format of our PDB-style files is described here.)
(The format of our PDB-style files is described here.)
Timeline for d1l1ye_:
- d1l1ye_ first appeared in SCOP 1.61
- d1l1ye_ appears in SCOPe 2.07
 View in 3D
View in 3D