Lineage for d5t25b_ (5t25 B:)
- Root: SCOPe 2.06

Class c: Alpha and beta proteins (a/b) [51349] (148 folds) 
Fold c.1: TIM beta/alpha-barrel [51350] (33 superfamilies)
contains parallel beta-sheet barrel, closed; n=8, S=8; strand order 12345678
the first seven superfamilies have similar phosphate-binding sites
Superfamily c.1.10: Aldolase [51569] (9 families) 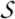
Common fold covers whole protein structure
Family c.1.10.1: Class I aldolase [51570] (13 proteins)
the catalytic lysine forms schiff-base intermediate with substrate
possible link between the aldolase superfamily and the phosphate-binding beta/alpha barrels
Protein Dihydrodipicolinate synthase [51574] (13 species) 
Species Escherichia coli [TaxId:585034] [323806] (2 PDB entries) 
Domain d5t25b_: 5t25 B: [330478]
Other proteins in same PDB: d5t25a2
automated match to d3du0a_
complexed with lys, na
Details for d5t25b_
PDB Entry: 5t25 (more details), 1.99 Å
PDB Description: kinetic, spectral and structural characterization of the slow binding inhibitor acetopyruvate with dihydrodipicolinate synthase from escherichia coli.
PDB Compounds: (B:) 4-hydroxy-tetrahydrodipicolinate synthaseSCOPe Domain Sequences for d5t25b_:
Sequence; same for both SEQRES and ATOM records: (download)
>d5t25b_ c.1.10.1 (B:) Dihydrodipicolinate synthase {Escherichia coli [TaxId: 585034]}
mftgsivaivtpmdekgnvcraslkklidyhvasgtsaivsvgttgesatlnhdehadvv
mmtldladgripviagtganataeaisltqrfndsgivgcltvtpyynrpsqeglyqhfk
aiaehtdlpqilynvpsrtgcdllpetvgrlakvkniigixeatgnltrvnqikelvsdd
fvllsgddasaldfmqlgghgvisvtanvaardmaqmcklaaeghfaearvinqrlmplh
nklfvepnpipvkwackelglvatdtlrlpmtpitdsgretvraalkhagll
SCOPe Domain Coordinates for d5t25b_:
Click to download the PDB-style file with coordinates for d5t25b_.
(The format of our PDB-style files is described here.)
(The format of our PDB-style files is described here.)
Timeline for d5t25b_:
- d5t25b_ is new in SCOPe 2.06-stable
- d5t25b_ appears in SCOPe 2.07
- d5t25b_ appears in the current release, SCOPe 2.08

 View in 3D
View in 3D

