Lineage for d5l3ue_ (5l3u E:)
- Root: SCOPe 2.08

Class d: Alpha and beta proteins (a+b) [53931] (396 folds) 
Fold d.92: Zincin-like [55485] (2 superfamilies)
contains mixed beta sheet with connection over free side of the sheet
Superfamily d.92.1: Metalloproteases ('zincins'), catalytic domain [55486] (18 families) 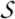

Family d.92.1.2: Thermolysin-like [55490] (5 proteins)
includes alpha-helical C-terminal domain characteristic for the family
Protein Thermolysin [63414] (3 species) 
Species Bacillus thermoproteolyticus [TaxId:1427] [55494] (191 PDB entries)
Uniprot P00800
Domain d5l3ue_: 5l3u E: [327390]
automated match to d1kkka_
complexed with 6ng, ca, dms, zn
Details for d5l3ue_
PDB Entry: 5l3u (more details), 1.23 Å
PDB Description: thermolysin in complex with jc149 (mpd cryo protectant)
PDB Compounds: (E:) thermolysinSCOPe Domain Sequences for d5l3ue_:
Sequence; same for both SEQRES and ATOM records: (download)
>d5l3ue_ d.92.1.2 (E:) Thermolysin {Bacillus thermoproteolyticus [TaxId: 1427]}
itgtstvgvgrgvlgdqkninttystyyylqdntrgngiftydakyrttlpgslwadadn
qffasydapavdahyyagvtydyyknvhnrlsydgnnaairssvhysqgynnafwngsqm
vygdgdgqtfiplsggidvvahelthavtdytagliyqnesgaineaisdifgtlvefya
nknpdweigedvytpgisgdslrsmsdpakygdpdhyskrytgtqdnggvhinsgiinka
aylisqggthygvsvvgigrdklgkifyraltqyltptsnfsqlraaavqsatdlygsts
qevasvkqafdavgvk
SCOPe Domain Coordinates for d5l3ue_:
Click to download the PDB-style file with coordinates for d5l3ue_.
(The format of our PDB-style files is described here.)
(The format of our PDB-style files is described here.)
Timeline for d5l3ue_:
- d5l3ue_ first appeared in SCOPe 2.06
- d5l3ue_ appears in SCOPe 2.07

 View in 3D
View in 3D