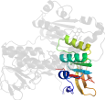
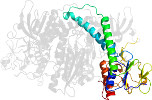
Lineage for d3sqpb1 (3sqp B:18-165,B:291-363)
- Root: SCOPe 2.06

Class c: Alpha and beta proteins (a/b) [51349] (148 folds) 
Fold c.3: FAD/NAD(P)-binding domain [51904] (1 superfamily)
core: 3 layers, b/b/a; central parallel beta-sheet of 5 strands, order 32145; top antiparallel beta-sheet of 3 strands, meander
Superfamily c.3.1: FAD/NAD(P)-binding domain [51905] (9 families) 

Family c.3.1.5: FAD/NAD-linked reductases, N-terminal and central domains [51943] (15 proteins)
duplication: both domains have similar folds and functions
most members of the family contain common C-terminal alpha+beta domain
Protein Glutathione reductase [51944] (3 species) 
Species Human (Homo sapiens) [TaxId:9606] [51945] (23 PDB entries) 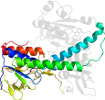
Domain d3sqpb1: 3sqp B:18-165,B:291-363 [261599]
Other proteins in same PDB: d3sqpa3, d3sqpb3
automated match to d3grsa1
protein/RNA complex; complexed with 3j8, fad, gol, so4
Details for d3sqpb1
PDB Entry: 3sqp (more details), 2.21 Å
PDB Description: Structure of human glutathione reductase complexed with pyocyanin, an agent with antimalarial activity
PDB Compounds: (B:) glutathione reductase, mitochondrialSCOPe Domain Sequences for d3sqpb1:
Sequence; same for both SEQRES and ATOM records: (download)
>d3sqpb1 c.3.1.5 (B:18-165,B:291-363) Glutathione reductase {Human (Homo sapiens) [TaxId: 9606]}
vasydylvigggsgglasarraaelgaraavveshklggtcvnvgcvpkkvmwntavhse
fmhdhadygfpscegkfnwrvikekrdayvsrlnaiyqnnltkshieiirghaaftsdpk
ptievsgkkytaphiliatggmpstpheXrvpntkdlslnklgiqtddkghiivdefqnt
nvkgiyavgdvcgkalltpvaiaagrklahrlfeykedskld
SCOPe Domain Coordinates for d3sqpb1:
Click to download the PDB-style file with coordinates for d3sqpb1.
(The format of our PDB-style files is described here.)
(The format of our PDB-style files is described here.)
Timeline for d3sqpb1:
- d3sqpb1 first appeared in SCOPe 2.04
- d3sqpb1 appears in SCOPe 2.05
- d3sqpb1 appears in SCOPe 2.07
- d3sqpb1 appears in the current release, SCOPe 2.08

 View in 3D
View in 3D